The Joint Nordic HTA Bodies – JNHB
The Nordic collaboration JNHB offers efficient and transparent joint health technology assessments of medicinal products in the five Nordic countries.
The collaborating HTA bodies are the Danish Medicines Council (DMC), the Finnish Medicines Agency (Fimea), Landspitali- University Hospital of Iceland, the Norwegian Medical Products Agency (NOMA) and the Dental and Pharmaceutical Benefits Agency (TLV) in Sweden.
A JNHB assessment adds two elements to national processes: The health technology developer (HTD) signs a Waiver of Confidentiality to allow information sharing, and submits the joint dossier and health economic model to TLV for distribution to the JNHB assessment team.
Assessments cover both relative effectiveness and health economics, supporting national decision-making in line with each country’s legal framework. While pricing and decisions are national responsibilities, JNHB supports these processes by delivering joint HTA reports based on shared evidence and established HTA methodology. The JNHB collaboration is not aiming for joint decisions on reimbursement or recommendations.
The figure below shows the organizations involved in joint HTA, negotiations, and decision-making.
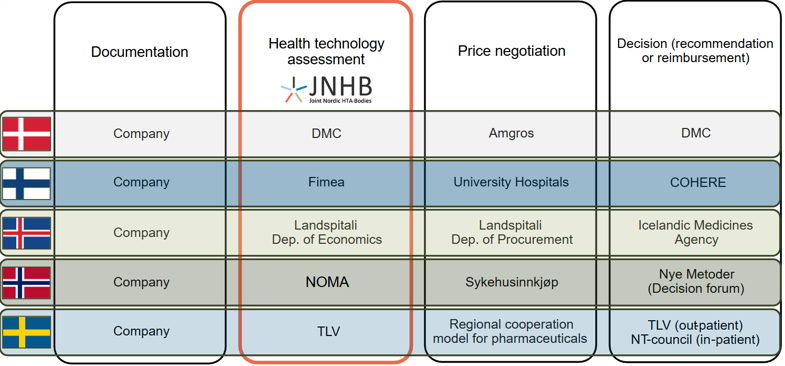
Find the fact sheet and read more:
Benefits of Nordic HTA Collaboration
By working together, JNHB aims at increasing efficiency and producing high quality assessment reports. Joint HTA allows the HTA bodies to share the different tasks of the assessments. Consolidating assessments into a single Nordic evaluation, supports equal and timely access to new cost-effective treatments for patients across the Nordic countries. A JNHB assessment can facilitate Nordic price negotiations for relevant products. However, the decisions on reimbursement and recommendation are national decisions made in each of the Nordic countries.
There are several key benefits from joint Nordic HTA of medicinal products.
Read more about key benefits here:
The main benefits for patients are:
Simultaneous assessments: Assessing new medicines jointly in the Nordic countries makes assessment reports accessible to Nordic decision makers simultaneously. This enables patients across the Nordic countries to access new treatments within the same time frame.
Harmonised approach to evaluation: In JNHB, assessments are made on a common evidence base and conclusions are summarised in a joint report. This ensures a harmonised approach to HTA and maintains high quality assessments that are professionally sound.
Decisions based on the same report: When decision makers in the Nordic countries base their decisions on a JNHB report, patients can trust that decisions are based on conclusions supported by all Nordic HTA bodies. This builds trust in decisions.
Short timelines: Short timelines support that new treatments can reach Nordic patients sooner. Working together enables Nordic HTA bodies to distribute tasks and share resources. Consequently, collaboration increases efficiency.
Potential for patients with rare diseases: Patients with rare diseases may face unequal access to treatments. Conducting a joint Nordic assessment for orphan products is one way to diminish differences in access for patients with rare diseases across the Nordic region.
There are also benefits for the Health Technology Developer. Resource sharing applies not only to HTA-bodies but also to companies involved in the process. HTDs benefit from a single, streamlined application process with one dossier and one point of contact, avoiding multiple parallel national procedures with differing requirements and timelines. Efficient procedures and short timelines for assessment should also benefit the HTD, as their new products will get the possibility to be available at the Nordic market earlier – when offered at a cost-effective price.
Memorandum of Understanding
The terms of the cooperation are clarified in the Memorandum of Understanding, originally signed in September 2017 and renewed in June 2020. The memorandum was renewed again when Denmark joined the collaboration in May 2023 and when Iceland joined in April 2024.

